A Vaccine that Targets Resistant Tumors by Dual T cell plus NK cell Attack
See manuscript in Nature (2022)
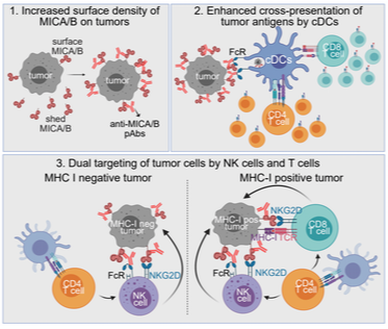
Most cancer vaccines target peptide antigens, necessitating personalization due to the vast inter-individual diversity of MHC molecules that present peptides to T cells. Furthermore, tumors frequently escape T cell-mediated immunity by mechanisms that interfere with peptide presentation. We developed a cancer vaccine that induces a coordinated attack by diverse T cell and NK cell populations. The vaccine targets the MICA and MICB (MICA/B) stress proteins expressed by many human cancers due to DNA damage. MICA/B serve as ligands for the activating NKG2D receptor on T cells and NK cells, but tumors evade immune recognition by proteolytic MICA/B cleavage. Vaccine-induced antibodies increase the density of MICA/B proteins on the surface of tumor cells by inhibiting proteolytic shedding, enhance presentation of tumor antigens by dendritic cells to T cells and augment the cytotoxic function of NK cells. Importantly, this vaccine maintains efficacy against MHC-I deficient tumors resistant to cytotoxic T cells through the coordinated action of NK cells and CD4 T cells. The vaccine is also efficacious in a clinically important setting: immunization following surgical removal of primary, highly metastatic tumors inhibits the later outgrowth of metastases. This vaccine design enables protective immunity even against tumors with common escape mutations.
See manuscript in Nature (2022)
Most cancer vaccines target peptide antigens, necessitating personalization due to the vast inter-individual diversity of MHC molecules that present peptides to T cells. Furthermore, tumors frequently escape T cell-mediated immunity by mechanisms that interfere with peptide presentation. We developed a cancer vaccine that induces a coordinated attack by diverse T cell and NK cell populations. The vaccine targets the MICA and MICB (MICA/B) stress proteins expressed by many human cancers due to DNA damage. MICA/B serve as ligands for the activating NKG2D receptor on T cells and NK cells, but tumors evade immune recognition by proteolytic MICA/B cleavage. Vaccine-induced antibodies increase the density of MICA/B proteins on the surface of tumor cells by inhibiting proteolytic shedding, enhance presentation of tumor antigens by dendritic cells to T cells and augment the cytotoxic function of NK cells. Importantly, this vaccine maintains efficacy against MHC-I deficient tumors resistant to cytotoxic T cells through the coordinated action of NK cells and CD4 T cells. The vaccine is also efficacious in a clinically important setting: immunization following surgical removal of primary, highly metastatic tumors inhibits the later outgrowth of metastases. This vaccine design enables protective immunity even against tumors with common escape mutations.
A cancer vaccine that retains efficacy against MHC-I negative tumor cells through a coordinated response by CD4 T cells and NK cells.
Induction of anti-tumor immunity by NK cells and T cells
See manuscripts in Science (2018), JCI Insight (2019), Cancer Immunology Research (2020)
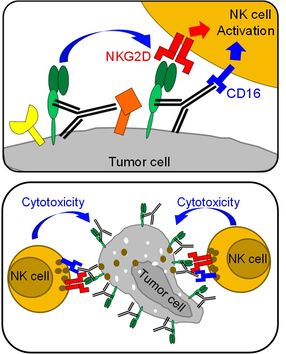
MICA and MICB (MICA/B) are expressed by many human cancers due to cellular stress and tag cells for elimination by cytotoxic lymphocytes through NKG2D receptor activation. However, tumors evade this immune recognition pathway through proteolytic shedding of MICA and MICB proteins. We rationally designed antibodies targeting the MICA α3 domain, the site of proteolytic shedding, and found that these antibodies prevented loss of cell surface MICA/B by human cancer cells.
These antibodies inhibited tumor growth in multiple fully immuno-competent mouse models and also reduced human melanoma metastases in a humanized mouse model. Anti-tumor immunity was mediated mainly by NK cells through activation of NKG2D and CD16 Fc receptors. This novel approach prevents the loss of important immunostimulatory ligands by human cancers and reactivates antitumor immunity (Science 2018).
See manuscripts in Science (2018), JCI Insight (2019), Cancer Immunology Research (2020)
MICA and MICB (MICA/B) are expressed by many human cancers due to cellular stress and tag cells for elimination by cytotoxic lymphocytes through NKG2D receptor activation. However, tumors evade this immune recognition pathway through proteolytic shedding of MICA and MICB proteins. We rationally designed antibodies targeting the MICA α3 domain, the site of proteolytic shedding, and found that these antibodies prevented loss of cell surface MICA/B by human cancer cells.
These antibodies inhibited tumor growth in multiple fully immuno-competent mouse models and also reduced human melanoma metastases in a humanized mouse model. Anti-tumor immunity was mediated mainly by NK cells through activation of NKG2D and CD16 Fc receptors. This novel approach prevents the loss of important immunostimulatory ligands by human cancers and reactivates antitumor immunity (Science 2018).
MICA antibodies block proteolytic shedding and enable activation of NK cells through the NKG2D and CD16 receptors. This results in potent NK cell-mediated tumor immunity.
Tumors frequently evade T cell-mediated attack by downregulation or loss of MHC class I expression (such as B2M mutation) or loss of IFNg signaling (such as JAK1 mutation). We showed that metastases with B2M or JAK1 mutations could be targeted by NK cells following treatment with a mAb that blocks MICA/B shedding (Cancer Immunology Research 2020). We also showed that a FDA approved HDAC inhibitor acted synergistically with this MICA/B mAb. The HDAC inhibitor enhanced MICA/B transcription while the MICA/B mAb stabilized the synthesized protein on the cell surface.
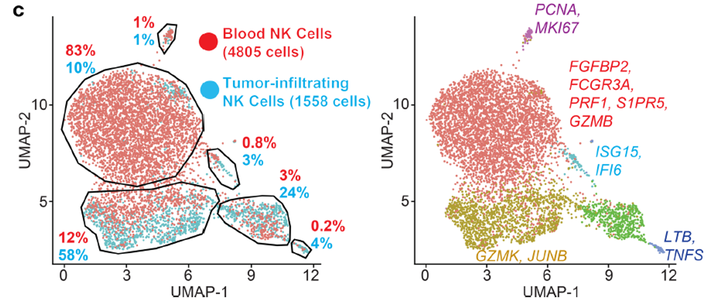
We are also performing single-cell RNA-seq analysis of NK cells isolated from human melanoma metastases to discover novel targets for NK cell and T cell-mediated tumor immunity. These studies show major differences in the transcriptional programs of tumor-infiltrating compared with circulating NK cells (JCI Insight 2019).
We are also performing single-cell RNA-seq analysis of NK cells isolated from human melanoma metastases to discover novel targets for NK cell and T cell-mediated tumor immunity. These studies show major differences in the transcriptional programs of tumor-infiltrating compared with circulating NK cells (JCI Insight 2019).
Striking differences in the gene expression programs of melanoma-infiltrating NK cells and blood NK cells from the same patient.
We are now investigating approaches that enable induction of a coordinated immune attack by CD8 T cells and NK cells. Given that these two cytotoxic lymphocyte populations recognize tumors by different mechanisms, a combined T cell and NK cell attack may inhibit the outgrowth of resistance mutants.
Badrinath, S., Dellacherie, M. O., Li, A., Zheng, S., Zhang, X., Sobral, M., Pyrdol, J. W., Smith, K. L.,Lu, Y.,Haag, S., Ijaz, H., Connor-Stroud, F., Kaisho, T., Dranoff, G., Yuan, G. C., Mooney, D. J., and Wucherpfennig,K. W. (2022) A vaccine targeting resistant tumors by dual T cell plus NK cell attack.Nature606, 992-998
Ferrari de Andrade, L., Tay, R. E., Pan, D., Luoma, A. M., Ito, Y., Badrinath, S., Tsoucas, D., Franz, B., May, K. F., Jr., Harvey, C. J., Kobold, S., Pyrdol, J. W., Yoon, C., Yuan, G. C., Hodi, F. S., Dranoff, G., and Wucherpfennig, K. W. (2018) Antibody-mediated inhibition of MICA and MICB shedding promotes NK cell-driven tumor immunity. Science 359, 1537-1542
Ferrari de Andrade, L., Kumar, S., Luoma, A. M., Ito, Y., Alves da Silva, P. H., Pan, D., Pyrdol, J. W., Yoon, C. H., and Wucherpfennig, K. W. (2020) Inhibition of MICA and MICB shedding elicits NK-cell-mediated immunity against tumors resistant to cytotoxic T cells. Cancer Immunology Research 8, 769-780
de Andrade, L. F., Lu, Y., Luoma, A., Ito, Y., Pan, D., Pyrdol, J. W., Yoon, C. H., Yuan, G. C., and Wucherpfennig, K. W. (2019) Discovery of specialized NK cell populations infiltrating human melanoma metastases. JCI Insight 4, e133103
Badrinath, S., Dellacherie, M. O., Li, A., Zheng, S., Zhang, X., Sobral, M., Pyrdol, J. W., Smith, K. L.,Lu, Y.,Haag, S., Ijaz, H., Connor-Stroud, F., Kaisho, T., Dranoff, G., Yuan, G. C., Mooney, D. J., and Wucherpfennig,K. W. (2022) A vaccine targeting resistant tumors by dual T cell plus NK cell attack.Nature606, 992-998
Ferrari de Andrade, L., Tay, R. E., Pan, D., Luoma, A. M., Ito, Y., Badrinath, S., Tsoucas, D., Franz, B., May, K. F., Jr., Harvey, C. J., Kobold, S., Pyrdol, J. W., Yoon, C., Yuan, G. C., Hodi, F. S., Dranoff, G., and Wucherpfennig, K. W. (2018) Antibody-mediated inhibition of MICA and MICB shedding promotes NK cell-driven tumor immunity. Science 359, 1537-1542
Ferrari de Andrade, L., Kumar, S., Luoma, A. M., Ito, Y., Alves da Silva, P. H., Pan, D., Pyrdol, J. W., Yoon, C. H., and Wucherpfennig, K. W. (2020) Inhibition of MICA and MICB shedding elicits NK-cell-mediated immunity against tumors resistant to cytotoxic T cells. Cancer Immunology Research 8, 769-780
de Andrade, L. F., Lu, Y., Luoma, A., Ito, Y., Pan, D., Pyrdol, J. W., Yoon, C. H., Yuan, G. C., and Wucherpfennig, K. W. (2019) Discovery of specialized NK cell populations infiltrating human melanoma metastases. JCI Insight 4, e133103